How They Work, Where They're Used, and How to Select Them
Connection interfaces are among the most overlooked contamination vectors in clinical environments. Antimicrobial connectors embed passive microbial suppression directly into the component — reducing risk at high-touch junction points in anesthesia, respiratory, fluid management, and veterinary systems without altering clinical workflow.
What Are Antimicrobial Connectors and Why Do They Matter in Clinical Systems?
Antimicrobial connectors are interface components designed to mechanically join medical tubing, devices, or systems while actively inhibiting microbial growth on contact surfaces. These connectors incorporate materials or coatings that suppress bacteria, fungi, and other pathogens, reducing contamination risk at high-touch connection points.
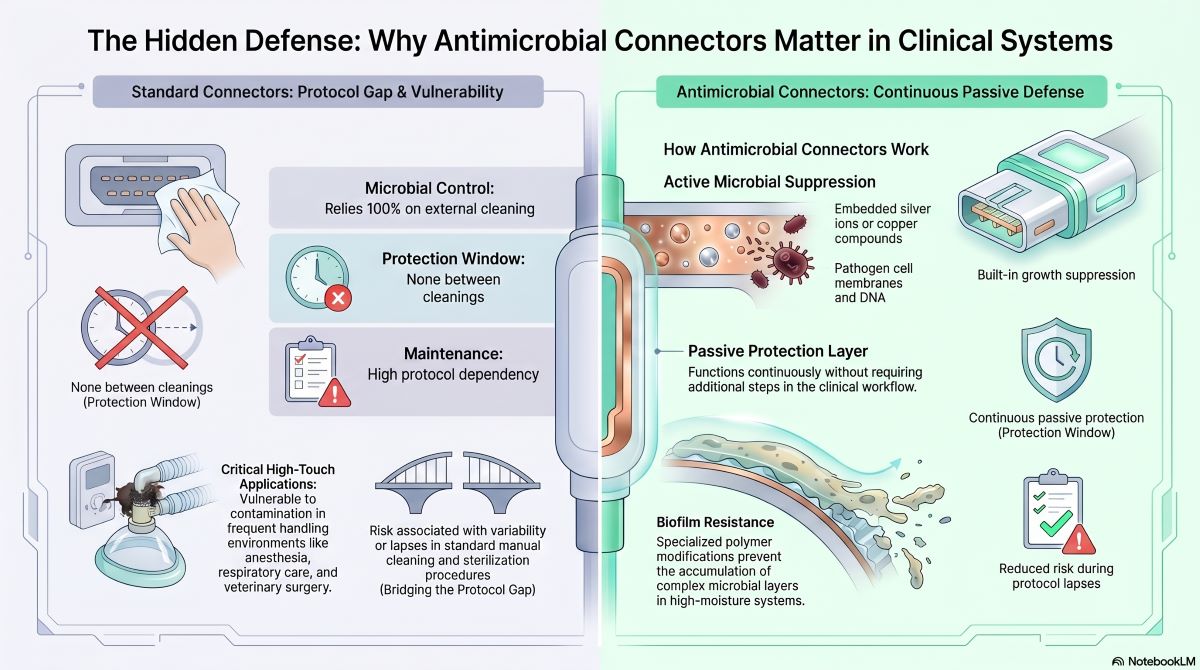
Connection interfaces are one of the most overlooked vectors of contamination in clinical environments. Fluids, aerosols, and repeated handling create conditions where microorganisms can accumulate — particularly in systems involving respiratory care, anesthesia delivery, and fluid management. Standard connectors rely entirely on cleaning protocols for infection control. Antimicrobial connectors introduce an additional layer of passive protection that functions continuously between cleaning cycles.
The relevance of antimicrobial connectors increases wherever connections are repeatedly made and broken. In intensive care units, surgical suites, and veterinary operating rooms, even small reductions in microbial load at junction points can meaningfully lower cross-contamination risk over time.
How Antimicrobial Connectors Reduce Microbial Contamination at the Source
Antimicrobial connectors reduce contamination by interrupting microbial survival and reproduction directly on the connector surface. The mechanism depends on the material science embedded within the connector — and it operates without any user intervention.
Most antimicrobial connectors operate through one or more of the following approaches:
- Embedded antimicrobial agents such as silver ions or copper compounds that disrupt microbial cell membranes
- Surface coatings that create inhospitable environments for microbial adhesion
- Polymer modifications that prevent biofilm formation throughout the material
- Hydrophobic surface treatments that reduce moisture retention, limiting microbial viability
- Contact-killing technologies that deactivate microbes upon surface interaction
An important clinical distinction: antimicrobial connectors do not create sterility — they reduce microbial load accumulation over time. This passive protection is particularly valuable during periods between handling or in emergency situations where ideal sterilization workflows may be disrupted.
Where Antimicrobial Connectors Are Used Across Medical and Veterinary Applications
Antimicrobial connectors are deployed in systems where fluid or gas transfer intersects with repeated handling and elevated contamination risk. Their value increases proportionally with connection frequency and biological exposure.
| Application Area | Why Antimicrobial Protection Is Relevant |
|---|---|
| Anesthesia delivery systems | Connectors linking breathing circuits, masks, and ventilators are exposed to respiratory secretions at every connection event |
| Ventilation and respiratory care | Ventilator circuit connectors are subject to condensation and biofilm formation over extended use periods |
| Infusion and fluid management | IV connectors and adapters face repeated access events, increasing cumulative contamination exposure |
| Suction and drainage systems | Connectors encounter biological materials that actively promote microbial growth |
| Veterinary surgical environments | Mixed-species exposure and variable sterilization workflows increase the need for passive contamination control |
| Diagnostic equipment interfaces | Reusable connectors benefit from reduced microbial carryover between patients or procedures |
In both human and veterinary medicine, connector-level contamination is often indirect but cumulative. Each connection event introduces an opportunity for microbial transfer — antimicrobial connectors reduce the impact of these repeated exposures across the full cycle of use.
The Difference Between Antimicrobial and Standard Connectors
The primary difference between antimicrobial and standard connectors is the presence of active microbial suppression built into the connector material or surface. Standard connectors function purely as mechanical interfaces — their safety depends entirely on environmental controls and procedural discipline.
| Feature | Standard Connectors | Antimicrobial Connectors |
|---|---|---|
| Microbial control | Relies entirely on external cleaning | Built-in suppression of microbial growth |
| Surface properties | Neutral — no active treatment | Treated or modified at material level |
| Biofilm resistance | Limited | Enhanced through material engineering |
| Maintenance dependency | High — fully protocol-dependent | Reduced — passive protection operates continuously |
| Protection between cleanings | None | Continuous passive protection |
| Cost profile | Lower upfront cost | Higher upfront; lower cumulative risk exposure |
This distinction does not eliminate the need for cleaning protocols. Rather, antimicrobial connectors reduce the consequences of lapses or variability in those protocols — shifting part of the infection control burden into the component itself.
Material Technologies Used in Antimicrobial Connectors
Antimicrobial effectiveness is determined by the materials and treatments integrated into the connector. The selection of these technologies directly affects durability, compatibility, and sustained performance over repeated use cycles.
| Technology | Mechanism | Key Consideration |
|---|---|---|
| Silver ion technology | Disrupts microbial DNA and cellular processes | Broad-spectrum effectiveness and good long-term stability |
| Copper-based compounds | Rapid surface-contact antimicrobial activity | Particularly effective against bacteria; material compatibility must be verified |
| Quaternary ammonium compounds (QACs) | Coating-based disruption of microbial cell membranes | Coating may degrade over time with repeated sterilization |
| Antimicrobial polymers | Agents embedded throughout the material, not just the surface | More durable than surface-only coatings; higher manufacturing cost |
| Photocatalytic coatings | Light-activated reactive species neutralize microbes | Effectiveness depends on light exposure; limited in enclosed systems |
Compatibility with sterilization methods must be verified for every technology selected. Repeated exposure to heat, steam, or chemical agents can degrade surface coatings or alter material properties — reducing antimicrobial performance over the connector's intended lifecycle.
Performance Considerations When Selecting Antimicrobial Connectors
Effective selection depends on aligning connector properties with the specific operational demands of the target environment. Not all antimicrobial connectors perform equally across different clinical settings or sterilization regimes.
Key Evaluation Criteria
- Spectrum of antimicrobial activity — the connector should target the bacteria, fungi, and pathogens relevant to the specific application environment
- Durability under sterilization — materials must maintain effectiveness after repeated cleaning and sterilization cycles, not just at baseline
- Mechanical integrity — antimicrobial treatments must not compromise connection strength, sealing performance, or dimensional tolerances
- Compatibility with existing systems — connectors must match standard sizes, fittings, and medical device interface requirements
- Resistance to biofilm formation — preventing biofilm development is critical in fluid-based or high-condensation systems
- Regulatory compliance — components must meet applicable medical device standards and certifications for the intended market
A connector that performs well in antimicrobial testing but degrades under sterilization cycles introduces risk rather than reducing it. Performance must be evaluated across the full operational lifecycle, not only under initial conditions.
Operational Impact: Infection Control, Maintenance, and Risk Reduction
Antimicrobial connectors reduce contamination risk by lowering microbial load at critical junction points, supporting broader infection control strategies. Their impact is incremental but cumulative — and most significant in high-frequency connection environments where standard protocol compliance is most variable.
From an operational perspective, these connectors:
- Reduce microbial transfer between connection events without additional workflow steps
- Lower dependence on perfect compliance with cleaning and sterilization protocols
- Extend intervals between contamination-related maintenance issues in fluid and gas systems
- Improve consistency across variable clinical workflows and shift-to-shift staff changes
- Support safer reuse of compatible systems where applicable under institutional guidelines
Antimicrobial connectors do not eliminate infection risk. They reduce the likelihood that connectors become reservoirs for microbial growth — and in systems where contamination compounds over time, this incremental reduction can meaningfully improve long-term outcomes.
Integration Challenges and How to Avoid Performance Gaps
Improper integration reduces the effectiveness of antimicrobial connectors. Selection without full system alignment introduces avoidable risks that can negate the performance benefits of the technology entirely.
Common Integration Issues
- Mismatched connector sizes leading to leakage, improper sealing, or mechanical failure under pressure
- Incompatible sterilization processes that degrade antimicrobial coatings or embedded materials over time
- Partial system upgrades that leave high-risk connection points untreated while others are replaced
- Overreliance on antimicrobial features without maintaining existing cleaning and sterilization protocols
Practical Mitigation Steps
- Standardize connector specifications consistently across all systems in a given clinical area
- Validate compatibility with all sterilization methods in use before full deployment
- Replace connectors consistently across complete critical pathways — not selectively at individual points
- Maintain existing infection control procedures without modification when introducing antimicrobial connectors
Effective implementation requires consistency. Isolated or partial use of antimicrobial connectors limits their system-wide impact.
Decision Framework for Selecting Antimicrobial Connectors
Selecting the correct antimicrobial connector requires balancing performance, compatibility, and operational constraints specific to the clinical environment. This structured evaluation ensures connectors are chosen based on operational need rather than generalized assumptions.
| Decision Factor | Priority Consideration |
|---|---|
| Clinical environment | ICU, surgical suite, veterinary OR, or diagnostic setting — each has different exposure profiles |
| Frequency of handling | High-touch systems justify greater investment in antimicrobial properties than low-interaction systems |
| Exposure type | Fluid-contact, aerosol, or mixed-environment systems have different biofilm and contamination risk profiles |
| Sterilization method | Heat, chemical, or single-use protocols must be compatible with the connector's antimicrobial technology |
| System compatibility | New connectors must standardize with existing connector specifications across the facility |
| Risk tolerance | Higher-acuity environments with immunocompromised populations warrant stricter infection control component specifications |
BOMImed Antimicrobial Connectors: Designed for Clinical and Veterinary Demands
BOMImed antimicrobial connectors are designed to integrate seamlessly into existing medical and veterinary systems while supporting contamination control at the connection level. The focus is on combining mechanical reliability with material-level microbial resistance — without adding workflow complexity.
These connectors are engineered to:
- Maintain compatibility with standard medical fittings and adapters across common system configurations
- Withstand repeated sterilization and clinical use cycles without loss of mechanical or antimicrobial performance
- Provide consistent antimicrobial protection across operational conditions in both human and veterinary clinical environments
- Support applications ranging from anesthesia circuit interfaces to fluid management system connections
Within environments where connector reliability and infection control intersect, BOMImed connectors are positioned as functional system components — not specialized add-ons. Their value lies in reducing cumulative contamination risk without altering the workflows clinicians already depend on.
As part of BOMImed's broader portfolio of respiratory, anesthesia, and veterinary diagnostic equipment in Canada, antimicrobial connectors are supported by the same commitment to equipment reliability and clinical service that defines BOMImed's approach to medical supply.
Frequently Asked Questions: Antimicrobial Connectors in Medical Applications
What do antimicrobial connectors actually do?
Antimicrobial connectors inhibit the growth and reproduction of microorganisms on their surfaces, reducing contamination risk at connection points between medical tubing, devices, and systems. This protection is passive and continuous — it operates between cleaning cycles without any additional user action.
Are antimicrobial connectors necessary in all clinical systems?
They are most valuable in systems with frequent handling, fluid or aerosol exposure, or elevated infection control requirements — such as ICU ventilator circuits, anesthesia delivery systems, and IV access points. Lower-frequency or lower-risk connection interfaces may not justify the additional cost.
Do antimicrobial connectors eliminate bacteria completely?
No. Antimicrobial connectors reduce microbial growth and accumulation on the connector surface but do not create sterile conditions. They should be understood as a supplementary layer of contamination control, not a replacement for sterilization protocols.
Can antimicrobial connectors replace cleaning procedures?
They cannot replace cleaning or sterilization protocols and must be used alongside standard infection control practices. Their function is to reduce the consequences of variability or lapses in those protocols — not to eliminate the need for them.
How long do antimicrobial properties last?
Effectiveness depends on the specific material technology and usage conditions, particularly the frequency and type of sterilization exposure. Surface coatings may degrade over time, while embedded antimicrobial polymers and silver ion technologies tend to maintain performance longer across repeated use cycles.
Are antimicrobial connectors compatible with standard medical equipment?
Most are designed to match standard medical connector sizes and fittings, but compatibility should always be verified for the specific systems in use. Dimensional mismatches can compromise sealing integrity and negate the safety benefits of the antimicrobial properties.
Do antimicrobial connectors impact system performance?
Properly designed antimicrobial connectors maintain full mechanical and functional performance — including connection strength, sealing integrity, and dimensional tolerances — while adding passive microbial resistance. Antimicrobial treatments that compromise mechanical performance indicate a design or compatibility issue.
Where are antimicrobial connectors most commonly used in veterinary settings?
Veterinary applications include anesthesia machine breathing circuit connections, surgical suction systems, and fluid management interfaces. Mixed-species exposure environments and variable sterilization workflows in veterinary practice make passive contamination control particularly valuable compared to settings with more standardized human clinical protocols.
BOMImed Antimicrobial Connectors for Canadian Clinical Settings
BOMImed offers antimicrobial connectors designed for compatibility, durability, and passive infection control across medical and veterinary applications. Explore our respiratory and connector product range or contact our team for clinical support.
View Respiratory Products →