Portable spirometry machines enable clinical-grade lung function testing outside traditional lab settings
Portable spirometry machines provide accurate, repeatable measurements of lung function in environments where full pulmonary labs are not practical. Modern devices are engineered to deliver clinical-grade data while supporting remote monitoring, decentralized care, and point-of-care diagnostics.
The shift toward portable systems reflects operational realities. Clinics require faster throughput, home monitoring supports chronic disease management, and providers increasingly rely on distributed testing models that maintain diagnostic integrity without centralized infrastructure.
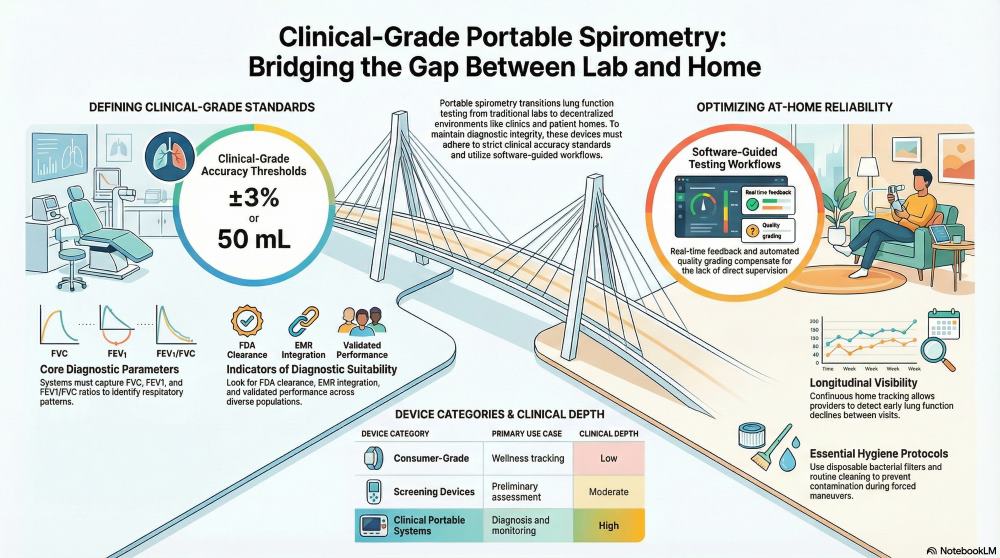
Clinical-grade performance depends on accuracy standards, calibration integrity, and repeatability
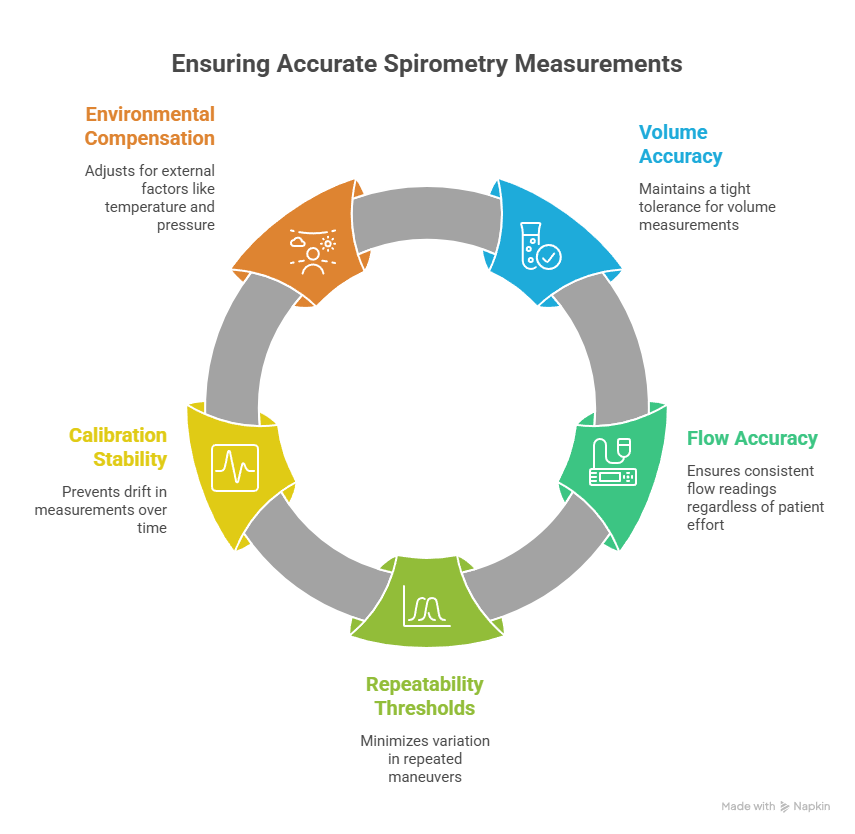
Clinical-grade spirometry is defined by adherence to strict measurement standards rather than device size or portability. Accuracy, calibration stability, and reproducibility determine whether a portable spirometry machine can be used for diagnostic or monitoring purposes.
Key performance criteria include:
- Volume accuracy: Typically within ±3% or 50 mL, whichever is greater
- Flow accuracy: Consistent measurement across variable patient effort levels
- Repeatability thresholds: Minimal variation across multiple forced expiratory maneuvers
- Calibration stability: Reduced drift over time or between sessions
- Environmental compensation: Adjustments for temperature, pressure, and humidity
Devices that meet these thresholds can support clinical decisions rather than serve as screening tools only.
Remote testing environments require different device capabilities than in-clinic spirometry
Remote and decentralized testing introduces variables that do not exist in controlled clinical environments. Portable spirometry machines must compensate for inconsistent technique, varying patient compliance, and limited supervision.
Device design priorities for remote use
Remote-ready spirometers prioritize usability without compromising measurement validity:
- Guided testing workflows embedded in software
- Real-time feedback on patient effort and technique
- Automated quality grading of maneuvers
- Minimal setup and calibration requirements
- Wireless data transmission to clinical systems
Operational differences between home and office testing
| Factor | Home Spirometry | Office Spirometry |
| Supervision | Self-administered or remote guidance | Direct clinical supervision |
| Environmental control | Variable | Controlled |
| Patient technique | Less consistent | Clinician-guided |
| Device complexity | Simplified interfaces | More advanced configurations |
| Data validation | Software-assisted | Clinician-reviewed in real time |
Devices designed for both environments must bridge these differences without degrading data reliability.
Home spirometry introduces clinical advantages when paired with structured monitoring protocols
Home spirometry enables continuous tracking of lung function, which is particularly valuable for chronic respiratory conditions. The primary advantage is longitudinal visibility rather than single-point diagnostics.
Structured home spirometry programs allow providers to:
- Detect early declines in lung function
- Monitor treatment response over time
- Reduce unnecessary in-person visits
- Support post-operative or post-discharge recovery
- Improve patient engagement in disease management
The clinical value depends on consistent testing frequency and adherence to standardized measurement protocols.
Not all portable spirometry machines are suitable for diagnostic use
Many devices marketed for portability lack the precision required for clinical decision-making. The distinction lies in regulatory classification, validation studies, and adherence to recognized spirometry standards.
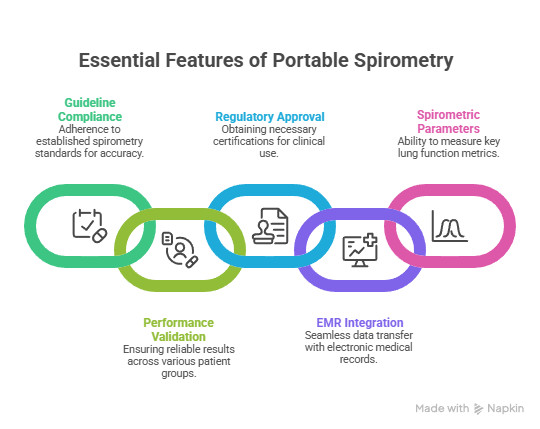
Indicators of diagnostic-grade capability
A portable spirometry machine suitable for clinical use typically demonstrates:
- Compliance with established spirometry guidelines
- Validated performance across diverse patient populations
- FDA clearance or equivalent regulatory approval
- Integration with electronic medical records (EMR/EHR)
- Ability to perform full spirometric parameters (FVC, FEV1, FEV1/FVC ratio)
Devices lacking these characteristics are better suited for wellness tracking or preliminary screening.
Spirometry test at home accuracy depends heavily on patient technique and device feedback systems
At-home spirometry introduces variability that can affect test reliability. Unlike clinical settings, patients perform maneuvers without direct supervision, increasing the risk of suboptimal effort or incorrect technique.
Effective systems mitigate these risks through:
- Real-time coaching prompts
- Visual or audio feedback during exhalation
- Automated rejection of poor-quality attempts
- Step-by-step guided workflows
Devices that fail to provide active guidance tend to produce inconsistent data, limiting their clinical usefulness.
Can you do a spirometry test at home with the same reliability as in-clinic testing
Home spirometry can achieve comparable reliability when device quality and patient adherence align with clinical protocols. However, consistency depends on structured implementation rather than device capability alone.
Conditions required for reliable at-home spirometry
- Patients are trained on proper technique
- Devices include automated quality control features
- Testing is performed under consistent conditions
- Results are reviewed or monitored by clinicians
- Data transmission is secure and uninterrupted
Without these safeguards, variability increases and diagnostic confidence decreases.
Key spirometric measurements determine whether a portable device meets clinical expectations
Spirometry machines must capture a range of parameters to support meaningful clinical interpretation. Devices that measure only basic metrics may not provide sufficient diagnostic depth.
Core measurements required
- Forced Vital Capacity (FVC): Total exhaled volume
- Forced Expiratory Volume in 1 second (FEV1): Volume exhaled in the first second
- FEV1/FVC ratio: Indicator of obstructive vs restrictive patterns
- Peak Expiratory Flow (PEF): Maximum speed of exhalation
Extended capabilities in advanced devices
- Flow-volume loop visualization
- Bronchodilator response tracking
- Trend analysis over time
- Predictive value comparison based on demographics
Devices that include these capabilities provide more complete clinical insight.
Device selection should prioritize clinical workflow integration over feature density
The most effective portable spirometry machines align with how clinicians and patients actually use them. Feature-rich devices that disrupt workflow often lead to underutilization or inconsistent testing.
Practical selection criteria
- Ease of use for both patients and clinicians
- Seamless integration with clinical software systems
- Minimal maintenance and calibration requirements
- Reliable connectivity for data transfer
- Clear result visualization for interpretation
Overly complex systems increase friction without improving outcomes.
Infection control and hygiene protocols are critical in portable spirometry deployment
Spirometry involves forced exhalation, which introduces contamination risks in both home and clinical settings. Proper hygiene protocols are essential to maintain safety and device longevity.
Standard hygiene practices
- Use of disposable mouthpieces or bacterial filters
- Routine cleaning of reusable components
- Avoidance of cross-patient device sharing without sterilization
- Proper storage to prevent environmental contamination
Devices designed with infection control in mind reduce operational risk.
Battery life, portability, and durability influence real-world usability
Portable spirometry machines must perform reliably across varying conditions. Battery performance and physical durability are often overlooked but directly impact usability.
Operational considerations
- Battery lifespan sufficient for multiple testing sessions
- Resistance to drops or environmental exposure
- Compact form factor for transport or home storage
- Quick startup time for on-demand testing
Devices that fail in these areas introduce operational inefficiencies.
Data management capabilities determine long-term clinical value
Spirometry data is most valuable when it can be stored, analyzed, and integrated into broader clinical systems. Isolated data limits longitudinal analysis and clinical decision-making.
Essential data features
- Cloud-based storage and access
- Integration with EMR/EHR systems
- Automated trend analysis
- Secure patient data handling
- Export capabilities for reporting
Devices that lack robust data infrastructure reduce their long-term utility.
Common limitations of portable spirometry machines must be understood before deployment
Portable spirometry introduces trade-offs that must be acknowledged. While convenience improves access, certain limitations can affect clinical interpretation.
Key limitations
- Increased variability in patient-administered tests
- Potential calibration drift in lower-end devices
- Reduced ability to supervise technique in real time
- Environmental factors affecting measurements
- Dependence on patient compliance
Understanding these constraints allows for appropriate use and interpretation.
Clinical use cases extend beyond chronic disease monitoring
Portable spirometry machines support a wide range of clinical applications beyond long-term respiratory conditions.
Common use cases
- Pre-operative lung function assessment
- Occupational health screening
- Post-infection recovery monitoring
- Clinical trial data collection
- Primary care respiratory evaluation
These applications benefit from rapid, accessible testing without full lab infrastructure.
Portable spirometry machine categories vary by intended use and technical capability
Not all portable devices serve the same purpose. Categorization helps clarify appropriate use cases and expectations.
Device categories
| Category | Primary Use Case | Clinical Depth |
| Consumer-grade | Wellness tracking | Low |
| Screening devices | Preliminary assessment | Moderate |
| Clinical portable systems | Diagnosis and monitoring | High |
| Integrated telehealth units | Remote patient management | High with connectivity |
Selecting the correct category prevents misuse and ensures appropriate expectations.
FAQ: Portable Spirometry and Home Testing
Can you do a spirometry test at home safely
Yes, home spirometry can be performed safely when using devices with proper hygiene controls and guided testing features.
How accurate is a portable spirometry machine compared to lab equipment
Clinical-grade portable devices can achieve comparable accuracy when properly calibrated and used according to protocol.
What conditions can home spirometry help monitor
Home spirometry is commonly used for asthma, COPD, post-surgical recovery, and other chronic respiratory conditions.
How often should spirometry be performed at home
Frequency depends on the condition being monitored, but consistent, scheduled testing is required for meaningful trend analysis.
Do portable spirometry machines require calibration
Some devices require periodic calibration, while others are factory-calibrated with minimal maintenance requirements.
What is the most important measurement in spirometry
FEV1 is often the most critical metric for assessing airflow limitation and disease progression.
Can spirometry detect early lung disease at home
Spirometry can identify early changes in lung function, but interpretation should be performed by a qualified clinician.