Incentive spirometry units are handheld respiratory devices designed to promote sustained maximal inspiration in patients at risk of postoperative pulmonary complications. The equipment provides visual feedback that encourages controlled, deep breathing to re-expand alveoli and restore lung function after anesthesia, surgery, or prolonged immobilization.
Incentive spirometry equipment is most commonly used following thoracic, abdominal, and upper airway procedures, where reduced inspiratory effort increases the likelihood of atelectasis. The device functions as a guided breathing tool rather than a passive oxygen-delivery system, making patient participation central to its effectiveness.
Most incentive spirometry units consist of a calibrated chamber, a flow or volume indicator, and a mouthpiece connected by flexible tubing. The design allows clinicians to quantify inspiratory capacity while reinforcing respiratory muscle engagement during recovery.
Why incentive spirometry matters in post-surgical pulmonary recovery
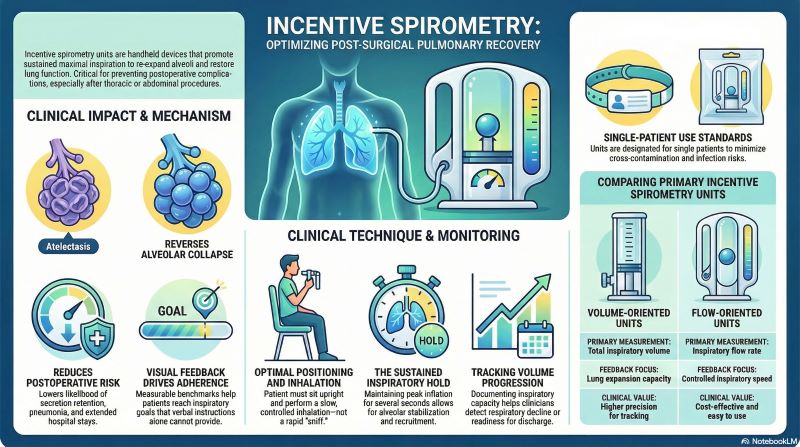
Incentive spirometry directly supports lung re-expansion and reduces the risk of atelectasis following surgery. General anesthesia, pain-related splinting, and supine positioning impair diaphragmatic movement and diminish tidal volume. Without intervention, these factors contribute to secretion retention, ventilation-perfusion mismatch, and pneumonia.
Postoperative pulmonary complications significantly increase hospital length of stay and resource utilization. Incentive spirometry equipment provides a structured method to restore inspiratory capacity, promote alveolar recruitment, and improve ventilation distribution.
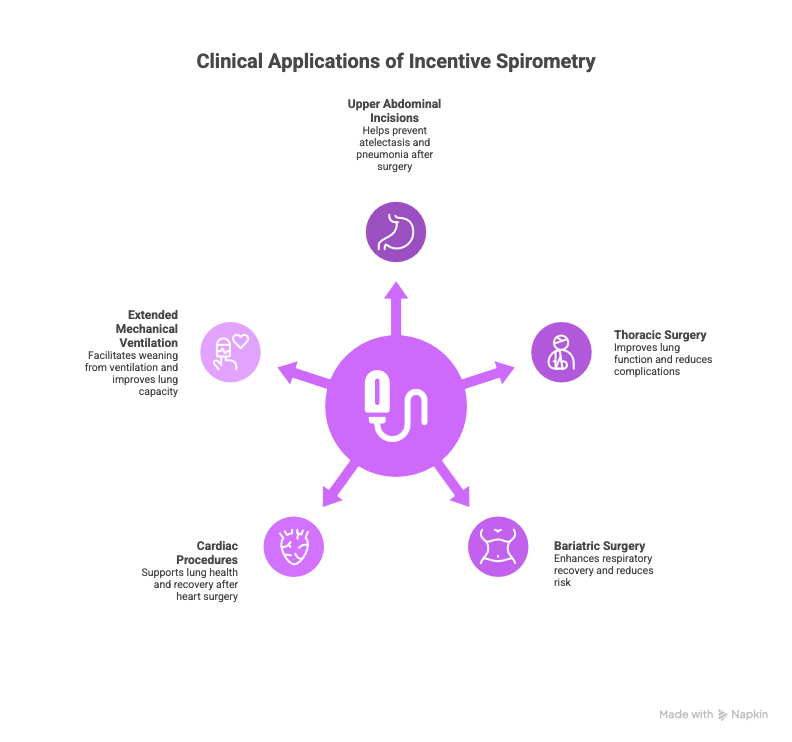
Clinical use is particularly relevant in procedures involving:
- Upper abdominal incisions
- Thoracic surgery
- Bariatric surgery
- Cardiac procedures
- Extended mechanical ventilation
By restoring deep breathing patterns early in recovery, incentive spirometry units reduce the progression from mild atelectasis to more severe pulmonary compromise.
What proper incentive spirometry technique looks like in clinical settings
Proper incentive spirometry technique requires slow, controlled inhalation to a predetermined target volume, followed by a brief inspiratory hold before relaxed exhalation. The patient must be seated upright or in a high Fowler’s position to allow optimal diaphragmatic expansion. Suboptimal positioning significantly reduces inspiratory capacity and compromises the therapeutic effect.
Clinical instruction typically emphasizes a smooth inhalation rather than a forceful breath. Rapid inspiration activates accessory muscles and reduces sustained alveolar recruitment. The inspiratory hold—generally several seconds—allows time for alveolar stabilization before exhalation.
Incorrect technique commonly includes:
- Short, shallow inhalations
- Rapid “sniffing” breaths
- Failure to hold the breath at peak inspiration
- Slouched or supine posture
- Irregular usage intervals
Direct supervision during early postoperative sessions improves long-term compliance and technique consistency.
Incentive spirometry units versus alternative lung expansion strategies
Incentive spirometry units differ from other postoperative pulmonary hygiene interventions in their emphasis on visual feedback and patient-driven inspiratory goals. Deep breathing exercises without equipment rely entirely on verbal instruction, which can reduce adherence and measurement accuracy.
Alternative methods include:
- Intermittent positive pressure breathing
- Continuous positive airway pressure (CPAP)
- Early mobilization protocols
- Directed coughing exercises
- Chest physiotherapy
Each method serves a distinct purpose. Incentive spirometry equipment is particularly effective in cooperative, spontaneously breathing patients who require structured lung re-expansion. Positive pressure systems are reserved for more severe or high-risk cases where independent inspiratory effort is insufficient.
In lower-risk surgical populations, early ambulation combined with deep breathing may achieve similar outcomes. However, incentive spirometry units offer measurable benchmarks that support standardized postoperative monitoring.
Clinical indications that warrant routine use of incentive spirometry equipment
Routine use of incentive spirometry equipment is indicated in patients with elevated risk of postoperative pulmonary compromise. Risk stratification considers surgical site, anesthesia duration, baseline pulmonary function, and comorbid conditions.
High-priority clinical scenarios include:
- Upper abdominal or thoracic surgery
- History of chronic obstructive pulmonary disease
- Obesity with restricted diaphragmatic movement
- Prolonged general anesthesia
- Reduced mobility following surgery
Preoperative inspiratory training may also be implemented in selected cases to improve baseline respiratory muscle performance before elective procedures.
Standardization of indications reduces variability across surgical units and improves postoperative pulmonary consistency.
What inspiratory volume targets actually represent
Inspiratory volume targets represent an estimate of the patient’s maximal inspiratory capacity under postoperative conditions. The goal is not to achieve preoperative pulmonary function immediately but to progressively restore lung expansion toward baseline.
Baseline measurement is often taken during the first supervised session. Subsequent sessions aim to maintain or gradually improve inspiratory volume.
Target interpretation must account for:
- Age-related lung capacity variation
- Body habitus
- Surgical pain limitation
- Preexisting pulmonary disease
- Sedation effects
Overly aggressive targets can cause patient frustration and poor adherence. Clinically appropriate targets emphasize gradual improvement rather than absolute numbers.
Postoperative timing and duration of incentive spirometry use
Incentive spirometry should begin as soon as the patient is alert and capable of following instructions after anesthesia. Early initiation mitigates the rapid onset of atelectasis that can occur within hours of surgery.
Usage generally continues until:
- The patient demonstrates stable inspiratory volumes
- Ambulation is consistent
- Pulmonary auscultation remains clear
- Oxygen saturation stabilizes without supplemental escalation
Extended use beyond the acute postoperative period may be warranted in patients with underlying pulmonary compromise. Duration must align with clinical assessment rather than fixed timeframes.
Risks and safety considerations associated with spirometry equipment
Incentive spirometry equipment carries minimal inherent risk when used appropriately. However, improper use or overexertion can lead to dizziness or hyperventilation in some patients.
Safety considerations include:
- Monitoring for lightheadedness
- Avoiding excessive breath-holding
- Ensuring clean, dry tubing
- Reinforcing correct inhalation speed
Patients with recent thoracic trauma or unstable hemodynamics require careful evaluation before use. Incentive spirometry units are not appropriate for patients unable to maintain airway protection or adequate inspiratory effort.
Measuring outcomes tied to incentive spirometry interventions
Measuring outcomes involves assessing both objective respiratory metrics and broader recovery indicators. Improvement in inspiratory capacity suggests effective alveolar recruitment and diaphragmatic activation.
Additional outcome markers include:
- Reduced incidence of postoperative atelectasis
- Stable oxygen saturation
- Decreased need for escalation to positive pressure therapy
- Shorter duration of respiratory monitoring
While incentive spirometry equipment alone does not determine postoperative success, it contributes to a comprehensive pulmonary management strategy. Documentation of inspiratory progression provides a measurable framework within multidisciplinary care pathways.
Economic and operational considerations in equipment selection
Equipment procurement decisions must balance cost efficiency with durability and clinical reliability. Disposable incentive spirometry units simplify infection control and reduce reprocessing burden. Reusable systems require structured cleaning protocols and may increase labor demands.
Operational considerations include:
- Unit cost relative to surgical volume
- Storage footprint
- Ease of bedside setup
- Compatibility with standardized postoperative order sets
- Staff familiarity and training requirements
Hospitals with high surgical throughput benefit from equipment that minimizes training complexity while maintaining measurement accuracy. Incentive spirometry units should integrate seamlessly into nursing workflow without adding unnecessary operational friction.
The relationship between pain management and spirometry performance
Effective pain control directly influences inspiratory capacity. Uncontrolled incisional pain leads to shallow breathing and reduced engagement with incentive spirometry equipment.
Multimodal analgesia supports spirometry performance by:
- Allowing deeper diaphragmatic movement
- Reducing splinting behavior
- Improving willingness to perform scheduled sessions
- Enhancing overall pulmonary effort
Respiratory therapists and nursing staff must coordinate with pain management teams to optimize spirometry outcomes. Equipment alone cannot compensate for inadequate analgesia.
Standardizing postoperative respiratory protocols across institutions
Standardized protocols improve consistency in the use of incentive spirometry units. Variability in instruction frequency or documentation weakens therapeutic impact.
A structured approach typically includes:
- Clear initiation criteria
- Defined usage intervals
- Volume tracking benchmarks
- Escalation pathways for declining performance
- Defined discontinuation criteria
Protocol alignment ensures that incentive spirometry equipment contributes meaningfully to broader recovery objectives rather than functioning as a passive bedside accessory.
The physiological mechanism behind sustained maximal inspiration
Sustained maximal inspiration reopens collapsed alveoli and improves surfactant distribution. Incentive spirometry units facilitate this process by encouraging slow, controlled inhalation to a targeted inspiratory volume.
The physiological effects include:
- Increased transpulmonary pressure
- Enhanced alveolar inflation
- Improved functional residual capacity
- Better ventilation-perfusion matching
- Reduced microatelectasis
Unlike rapid breathing exercises, incentive spirometry emphasizes inspiratory hold. Maintaining lung inflation for several seconds allows for more effective alveolar recruitment and stabilization.
The device’s visual feedback mechanism strengthens neuromuscular coordination between the diaphragm and accessory muscles. This engagement is critical after surgery, when shallow breathing patterns are common.
Volume-oriented versus flow-oriented incentive spirometry equipment
Volume-oriented incentive spirometry units measure the total volume of inspired air and display it numerically or through calibrated chambers. Flow-oriented units instead measure inspiratory flow rate using movable indicators or balls within vertical chambers.
The primary functional difference is measurement focus. Volume-based devices emphasize reaching a set inspiratory capacity, while flow-based equipment emphasizes steady inspiratory speed.
| Feature | Volume-Oriented Units | Flow-Oriented Units |
|---|---|---|
| Primary Measurement | Inspiratory volume | Inspiratory flow rate |
| Feedback Precision | Higher | Moderate |
| Training Focus | Lung expansion capacity | Controlled inspiratory effort |
| Common Use | Hospitals, surgical wards | General post-op care |
| Clinical Preference | Often preferred in structured protocols | Widely used for ease and cost-effectiveness |
Volume-oriented incentive spirometry equipment is generally favored when precise monitoring of inspiratory capacity is required. Flow-based units remain common due to simplicity and cost efficiency.
What determines the effectiveness of incentive spirometry equipment
Patient adherence and correct technique determine the success of incentive spirometry units. The device itself does not create lung expansion; it provides structured guidance for patient-driven inhalation.
Key determinants of effectiveness include:
- Adequate pain control
- Proper patient positioning
- Clear instruction and demonstration
- Regular usage intervals
- Monitoring by nursing staff
Pain from abdominal or thoracic incisions often limits inspiratory effort. Without multimodal analgesia, incentive spirometry performance declines significantly.
Equipment design also influences usability. Ergonomic mouthpieces, clearly visible indicators, and durable construction improve compliance and accuracy in high-volume clinical settings.
How incentive spirometry units integrate into enhanced recovery pathways
Enhanced Recovery After Surgery (ERAS) protocols emphasize early mobilization, optimized analgesia, and pulmonary hygiene. Incentive spirometry equipment fits within this framework as a low-risk, noninvasive intervention that supports respiratory stability.
In ERAS pathways, incentive spirometry is typically initiated in the post-anesthesia care unit and continued during inpatient recovery. The structured breathing schedule often includes multiple sessions per hour while awake.
When combined with ambulation and coughing exercises, incentive spirometry units reduce secretion pooling and improve diaphragmatic excursion. Integration into standardized postoperative order sets improves consistency of use.
Common misconceptions surrounding incentive spirometry devices
Incentive spirometry is often misunderstood as a replacement for oxygen therapy. The device does not deliver oxygen; it trains inspiratory effort and lung expansion.
Another misconception is that short, rapid inhalations provide similar benefit. Effective use requires slow, sustained inspiration followed by a brief inspiratory hold.
There is also variability in expectations. Incentive spirometry equipment reduces pulmonary complications when used correctly, but it does not eliminate risk in high-acuity surgical populations.
Proper education mitigates these misunderstandings and improves therapeutic consistency.
Infection control considerations for spirometry equipment in clinical settings
Incentive spirometry units are typically designated for single-patient use to minimize cross-contamination. Shared respiratory devices introduce infection control risk, particularly in postoperative wards with immunocompromised individuals.
Best practices include:
- Single-patient allocation
- Routine inspection for moisture accumulation
- Timely disposal after discharge
- Clear labeling to prevent equipment sharing
Hospitals must balance cost management with infection prevention. Disposable incentive spirometry equipment simplifies compliance and reduces sterilization burdens.
Equipment design features that matter in high-volume clinical environments
Durability and clarity of feedback are critical in institutional settings. Incentive spirometry units must withstand frequent handling while maintaining measurement accuracy.
Important design attributes include:
- Clearly marked volume calibrations
- Stable base or ergonomic grip
- Transparent chamber visibility
- Smooth, low-resistance airflow
- Replaceable or integrated tubing
Flow resistance consistency ensures predictable training across patient populations. Devices with unstable airflow compromise inspiratory effort assessment.
High-quality incentive spirometry equipment supports standardized respiratory therapy protocols across surgical departments.
Pediatric and specialized patient applications
Pediatric incentive spirometry units are scaled for lower inspiratory volumes and may incorporate visual elements to encourage engagement. Children recovering from surgery require age-appropriate calibration to avoid unrealistic targets.
Specialized applications extend to:
- Post-trauma recovery
- Chronic pulmonary disease exacerbations
- Preoperative respiratory conditioning
- Neuromuscular weakness rehabilitation
Device selection must align with inspiratory capacity and clinical objectives. Adult equipment used in pediatric populations reduces usability and measurement relevance.
When incentive spirometry may offer limited benefit
Incentive spirometry equipment may provide limited benefit in patients unable to follow instructions or generate adequate inspiratory effort. Severe cognitive impairment, advanced neuromuscular disorders, or uncontrolled pain reduce efficacy.
Mechanical ventilation-dependent patients require alternative lung expansion strategies. Additionally, some evidence suggests that mobilization and deep breathing exercises alone may provide comparable benefit in low-risk surgical populations.
Clinical judgment determines when incentive spirometry units are appropriate versus when alternative pulmonary hygiene strategies are indicated.
Role of BOMImed in supporting clinical respiratory equipment needs
Reliable sourcing of incentive spirometry units is essential for maintaining standardized postoperative protocols. BOMImed supplies clinical respiratory equipment designed to meet institutional quality and usability standards.
In high-demand healthcare environments, consistent device performance and availability support patient safety and operational efficiency. Incentive spirometry equipment must align with clinical workflow, infection control policies, and respiratory therapy objectives.
BOMImed’s approach focuses on durable, clinically appropriate equipment that integrates into hospital and surgical recovery settings without compromising respiratory care standards.
Documentation and monitoring in postoperative respiratory care
Tracking inspiratory volume progression provides objective insight into pulmonary recovery. Nursing staff often document baseline inspiratory capacity and monitor improvement over time.
Quantitative monitoring supports:
- Early detection of respiratory decline
- Evaluation of pain management adequacy
- Adjustment of respiratory therapy interventions
- Discharge readiness assessment
Volume-oriented incentive spirometry units facilitate more precise tracking compared to flow-based devices. Documentation strengthens accountability within postoperative recovery protocols.
People Also Ask – Incentive Spirometry Units
What are incentive spirometry units used for?
Incentive spirometry units are used to encourage deep breathing after surgery to prevent atelectasis and reduce pulmonary complications.
How often should incentive spirometry be performed after surgery?
Frequency depends on surgical protocol, but usage is commonly recommended multiple times per hour while the patient is awake.
What is the difference between flow and volume incentive spirometry?
Flow devices measure inspiratory speed, while volume devices measure the total air inhaled during a breath.
Can incentive spirometry replace oxygen therapy?
Incentive spirometry does not replace oxygen therapy; it improves lung expansion rather than delivering supplemental oxygen.
Are incentive spirometry units reusable?
Most units are designed for single-patient use to prevent cross-contamination in clinical settings.
Who should not use incentive spirometry equipment?
Patients unable to follow instructions or generate sufficient inspiratory effort may not benefit from incentive spirometry.
Does incentive spirometry reduce pneumonia risk?
Proper use can reduce atelectasis, which in turn lowers the likelihood of postoperative pneumonia in at-risk patients.