What “right” means in spirometry equipment selection
The right spirometry equipment is the system that produces repeatable, standards-aligned results with minimal operator variability while fitting the realities of your clinical workflow. Accuracy alone is not enough in real practice because most failures in spirometry are process failures: poor coaching, inconsistent acceptability checks, weak calibration discipline, and inadequate infection-control routines.
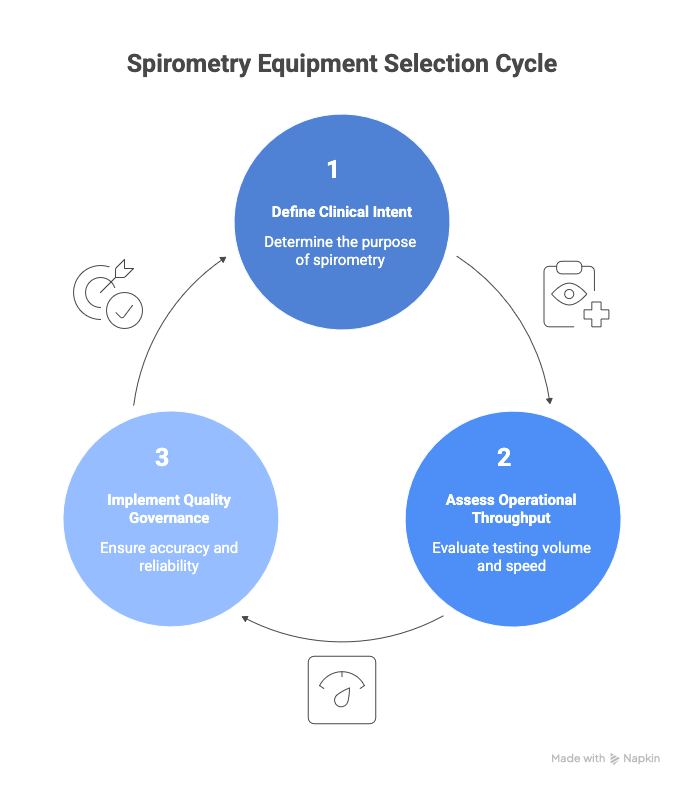
Selection decisions should be made around three forces that are always present but weighted differently by setting:
- Clinical intent (screening vs diagnosis vs longitudinal management)
- Operational throughput (how many tests, how fast, by whom)
- Quality governance (how you assure acceptability, repeatability, calibration, and reporting)
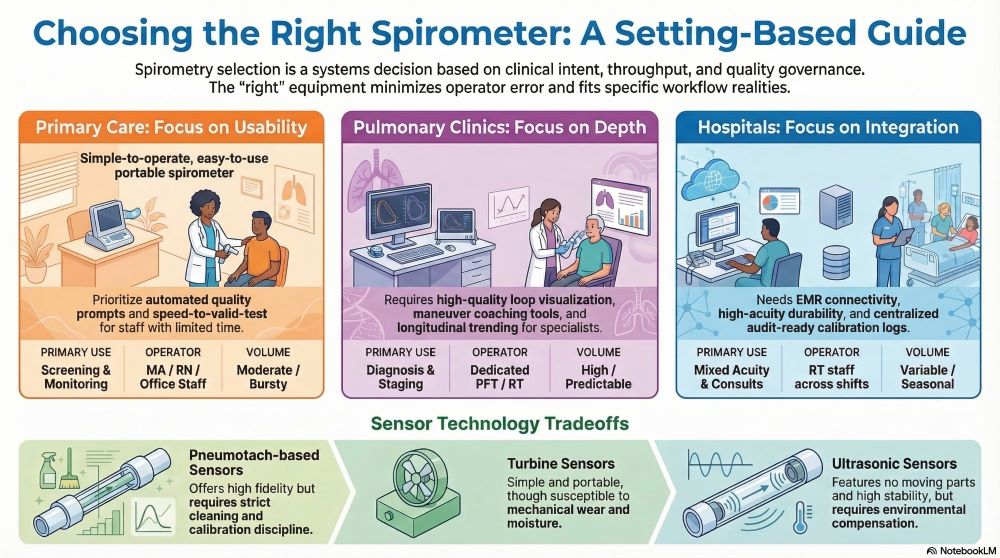
A primary care office generally needs speed, simplicity, and robust quality prompts. A pulmonary clinic needs tighter technical control, interpretive depth, and advanced maneuver support. A hospital needs multi-user durability, integration, and strict infection prevention in higher-acuity populations.
Which care setting you serve determines the minimum viable feature set
A spirometer that is “good” in a pulmonary function lab can be a poor choice in primary care if it slows throughput and increases operator error. A device that is “good enough” for screening can be a poor choice for specialty care if it cannot reliably support bronchodilator response workflows or high-quality flow-volume loop review.
Setting requirements at a glance
| Requirement Category | Primary Care | Pulmonary Clinic | Hospital / Health System |
|---|---|---|---|
| Primary use case | Screening, case-finding, monitoring | Diagnosis, staging, therapy response | Mixed acuity, inpatient/outpatient, consult-driven |
| Operator profile | MA/RN/office staff with limited time | Dedicated PFT staff or trained RT | RT staff across shifts; cross-coverage common |
| Essential quality controls | Strong automated acceptability prompts | Deep QC, loop review, maneuver coaching | QC + documentation + audit readiness |
| Volume / throughput | Moderate, bursty | High and predictable | Variable; can spike with seasonal respiratory load |
| Integration priority | Moderate | High | Very high |
| Infection control demands | High | High | Highest |
The correct choice is rarely “the most advanced device.” It is the device that makes it easiest to produce valid tests in your environment with your staff.
Define the clinical scope before you look at product specs
Spirometry equipment evaluation should start with a scope statement that clarifies which questions the test must answer in your organization. When the scope is unclear, teams buy for imagined future complexity and end up with a system that is underused, slow, or poorly maintained.
Scope questions that change equipment requirements:
- Will you use spirometry primarily for asthma and COPD assessment, or also for occupational surveillance, pre-op assessment, and therapy monitoring?
- Will you routinely perform pre/post bronchodilator testing?
- Do you need pediatric testing support (coaching tools, incentives, small mouthpieces, higher variability handling)?
- Do you require on-device interpretation aids, or will interpretation be centralized by specialists?
- Are you expected to meet lab-style quality governance, or office-based quality with automated prompts?
This scope becomes your minimum feature set and prevents decision drift during vendor discussions.
Standards alignment is a purchasing requirement, not a clinical afterthought
Spirometry quality depends on meeting modern performance expectations for device accuracy and routine verification. The practical implication is simple: your equipment choice must support a consistent calibration/verification routine and provide the data needed to defend test validity.
Equipment capabilities that support standards-aligned practice:
- Daily calibration verification support with a 3-L syringe workflow
- Clear reporting of acceptability and repeatability metrics
- Flow-volume loop display with sufficient resolution for review
- Automated quality prompts that guide staff during testing
- Audit-friendly storage of calibration logs and test metadata
If the device cannot make calibration verification and quality review routine, compliance will erode and results will become less dependable over time.
Sensor technology drives maintenance burden and long-term stability
Most clinical buyers focus on the user interface and miss the deeper driver of reliability: how flow and volume are measured. Sensor choices influence drift, cleaning needs, susceptibility to condensation, and the consistency of measurements across seasons and environments.
Common measurement approaches and practical tradeoffs
| Sensor Approach | Practical Strength | Practical Risk | Best-fit settings |
|---|---|---|---|
| Pneumotach-based (differential pressure) | High fidelity; widely accepted | Requires careful cleaning and calibration discipline; sensitive to contamination | Pulmonary clinics, hospitals |
| Turbine | Portable; simple; often lower cost | Mechanical wear; can be affected by moisture/secretions and resistance changes | Primary care, mobile programs |
| Ultrasonic | No moving parts; often stable over time | Environmental compensation needs; performance depends on implementation and workflow discipline | Primary care to specialty depending on model |
| Heated-wire / hot-wire (less common in routine office spirometry) | Sensitive flow detection | Can be more complex to maintain; contamination concerns | Specialized use cases |
A reliable decision framework is not “which sensor is best.” It is “which sensor is least likely to degrade quality under our staffing model and cleaning realities.”
Infection control is part of equipment selection, not just policy
Spirometry involves forced exhalation and close contact with patient interfaces, which raises the stakes for infection prevention. Device designs differ in how easily they support safe workflows with minimal shortcuts.
Selection criteria that materially affect infection control:
- Compatibility with single-use bacterial/viral filters without distorting measurements
- Clear guidance on whether calibration should be performed with the filter in place
- Disposable mouthpiece ecosystem availability and cost stability
- Ease of cleaning external surfaces between patients
- Ability to keep internal components protected from contamination through proper interface design
Primary care: prioritize usability, quality prompting, and speed-to-valid-test
Primary care succeeds with spirometry when the equipment reduces the two most common failure modes: inadequate coaching time and low confidence in test acceptability. The device should actively guide the operator toward valid maneuvers rather than merely recording attempts.
Primary care selection should emphasize:
- Fast setup with minimal consumables complexity
- Strong on-screen coaching cues and acceptability alerts
- Simple pre/post bronchodilator workflows when needed
- Automatic interpretation aids that support clinical decision-making without replacing it
- Lightweight reporting that can be documented quickly in the chart
Pulmonary clinics: prioritize maneuver control, interpretive depth, and auditability
Pulmonary clinics rely on spirometry as a diagnostic anchor and as a longitudinal tool for staging and treatment response. The equipment must support high technical quality at scale and preserve enough detail for expert interpretation.
Pulmonary clinic selection should emphasize:
- High-quality loop visualization and maneuver review
- Robust QC flags and easy access to acceptability details
- Efficient pre/post bronchodilator testing workflows
- Configurable predicted values and interpretive reference settings aligned to clinic policy
- Longitudinal trending tools that reduce interpretive variability over time
Hospitals: prioritize integration, multi-user resilience, and governance
Hospitals need spirometry capability that can handle varying acuity, multiple users across shifts, and strict governance expectations. Equipment in a hospital environment must be resilient to turnover, cross-coverage, and documentation requirements.
Hospital selection should emphasize:
- Seamless integration with EMR and respiratory documentation workflows
- Role-based access or user profiles to reduce variability and maintain accountability
- Centralized calibration and maintenance logging for compliance audits
- Durable hardware designed for frequent cleaning and frequent transport
- Vendor service responsiveness with clear uptime expectations
Choosing the Right Spirometry System – People Also Ask
What features matter most in spirometry equipment for primary care?
Strong automated quality prompts, fast setup, simple calibration verification, and EMR-friendly reporting matter more than advanced lab-style features.
Do pulmonary clinics need different spirometry equipment than primary care offices?
Pulmonary clinics typically need deeper QC tools, better loop review, standardized interpretive templates, and high-throughput workflow support.
How often should spirometry equipment be calibration-verified?
Calibration verification is commonly performed at least daily as part of routine quality assurance, using a consistent, documented process.
Are disposable filters necessary for spirometry testing?
Disposable bacterial/viral filters are commonly used to reduce cross-contamination risk, especially in higher-acuity environments and shared-device workflows.
What causes most “bad spirometry” results?
Poor coaching, inconsistent effort quality, and weak acceptability checks are the dominant causes, not the sensor technology itself.
Is portable spirometry reliable enough for clinical decisions?
Portable spirometry can be clinically reliable when it supports rigorous quality prompts, verification routines, and consistent operator technique.
How do hospitals ensure spirometry quality across multiple users?
Hospitals typically rely on standardized protocols, audit-ready logs, role-based training, and equipment that enforces QC steps consistently.
Spirometry programs that thrive treat equipment selection as a systems decision: measurement reliability, human coaching, infection control, data flow, and governance must all reinforce each other. As respiratory care becomes more longitudinal and digitally integrated, the most valuable spirometry equipment will be the systems that make high-quality testing easier to perform than low-quality testing—regardless of setting or staffing volatility.