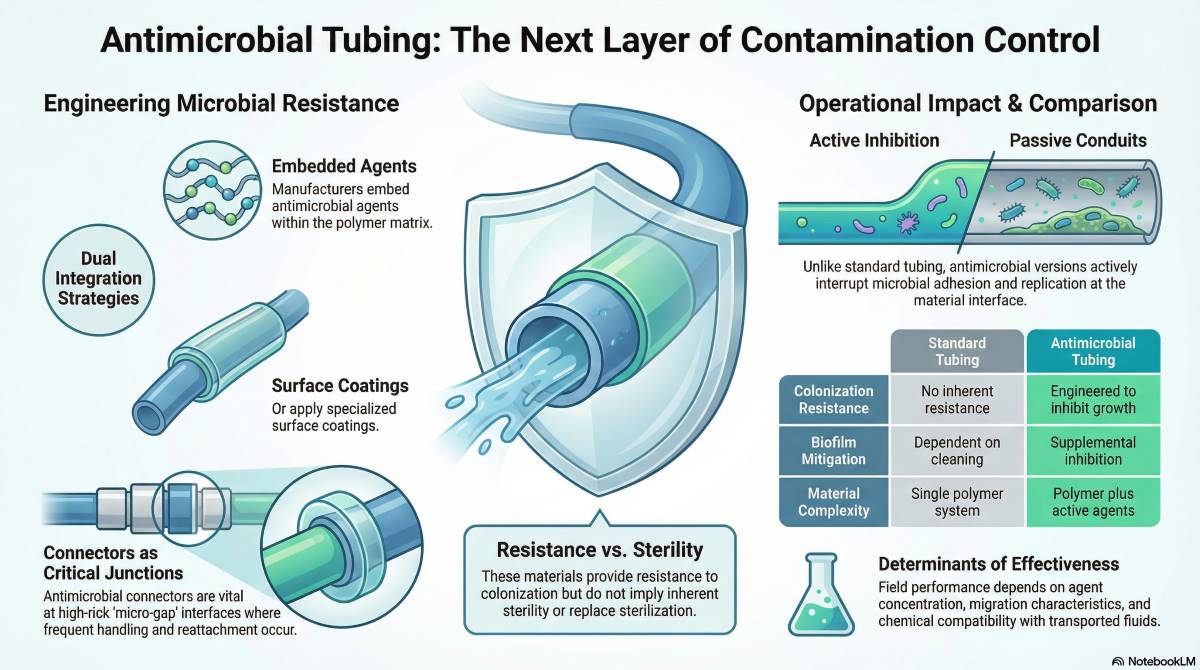
Anti microbial tubing refers to fluid-transfer tubing engineered to inhibit microbial growth on or within its material structure during operational use. The term does not imply sterility; it indicates resistance to colonization under defined environmental conditions. Performance depends on base polymer chemistry, additive integration, and exposure context.
In healthcare and laboratory settings, tubing functions as a transport pathway for fluids, gases, or biological samples. Surface colonization in these pathways can lead to biofilm formation, cross-contamination, and system degradation. Anti microbial tubing is designed to interrupt microbial adhesion, replication, or persistence at the material interface.
Two core strategies define the category:
- Incorporated antimicrobial agents embedded within the polymer matrix
- Surface-treated materials coated or bonded with antimicrobial compounds
Material selection influences efficacy, durability, chemical compatibility, and mechanical integrity. Silicone, PVC, polyurethane, and thermoplastic elastomers are commonly modified to support antimicrobial performance without compromising flexibility or transparency.
Regulatory classification and validation requirements vary depending on intended use. In critical care or surgical systems, anti microbial claims must align with controlled performance criteria. In general fluid handling applications, the objective centers on contamination risk reduction rather than infection control claims.
Why anti microbial connectors matter in contamination control
Anti microbial connectors reduce microbial transfer at junction points where tubing interfaces with devices, containers, or other fluid pathways. These junctions represent high-risk contamination sites because they are frequently handled, disconnected, and reattached.
Connectors introduce micro-gaps, crevices, and pressure differentials that can support microbial ingress. When connectors incorporate antimicrobial materials or surface technologies, the risk of microbial survival at these interfaces is lowered.
The operational importance of anti microbial connectors is most evident in systems requiring:
- Repeated fluid transfer
- Closed-loop integrity
- Minimal exposure to ambient contamination
- Extended dwell time
Connector geometry influences contamination risk as much as material composition. Luer locks, barbed fittings, quick-connect couplings, and custom molded assemblies must maintain sealing performance while integrating antimicrobial properties.
Material consistency between tubing and connectors is often overlooked. Mismatched materials can create differential wear patterns or micro-fractures, undermining antimicrobial intent. System-level design must consider compatibility rather than individual component performance.
Operational impact of anti microbial materials in fluid pathways
Anti microbial materials influence system performance beyond contamination reduction. They alter maintenance cycles, replacement intervals, and validation requirements.
In systems prone to biofilm development, antimicrobial materials can:
- Delay surface colonization
- Reduce microbial load accumulation
- Improve hygiene protocol effectiveness
- Lower downstream cleaning burden
- Support longer operational uptime
However, antimicrobial functionality does not eliminate the need for sterilization, disinfection, or proper handling procedures. It functions as a supplemental risk mitigation layer rather than a primary infection control measure.
In laboratory and veterinary environments, where frequent fluid exchanges occur, anti microbial tubing and connectors can reduce variability associated with environmental contamination. Stability in fluid pathways contributes to reproducibility and safety.
What determines anti microbial effectiveness in practice
Anti microbial effectiveness depends on material science, environmental exposure, and system design conditions. Laboratory efficacy does not automatically translate into field performance.
Key determinants include:
- Active agent concentration within the polymer
- Migration or leaching characteristics over time
- Surface wear and abrasion resistance
- Temperature and humidity exposure
- Chemical compatibility with transported fluids
For embedded antimicrobial agents, long-term durability is critical. Surface coatings may degrade with repeated sterilization or chemical exposure. In contrast, fully integrated polymer additives often provide more sustained performance but require careful validation.
Environmental load also influences results. High organic matter environments may shield microorganisms from contact-based antimicrobial surfaces. System flushing protocols and mechanical flow rates affect colonization potential.
Performance evaluation should consider real-world duty cycles rather than isolated laboratory testing.
Anti microbial tubing versus standard medical tubing
The primary distinction between anti microbial tubing and standard tubing is resistance to microbial growth at the material interface. Standard tubing provides a passive conduit; anti microbial tubing incorporates active or contact-based microbial inhibition.
A structured comparison clarifies functional differences:
| Attribute | Standard Tubing | Anti Microbial Tubing |
|---|---|---|
| Surface colonization resistance | No inherent resistance | Engineered to inhibit growth |
| Biofilm mitigation | Dependent on cleaning | Supplemental inhibition |
| Material complexity | Single polymer system | Polymer plus active agents |
| Cost profile | Lower | Higher |
| Validation requirements | Standard | Enhanced performance claims |
Mechanical properties may remain similar between the two categories. Flexibility, transparency, and pressure tolerance can be preserved with proper formulation.
Selection should align with contamination risk profile, not solely cost considerations.
Critical use environments where anti microbial connectors add value
Anti microbial connectors provide the greatest benefit in environments with repeated handling and extended system exposure. High-contact systems introduce consistent contamination risk at connection points.
Use cases include:
- Fluid sampling lines
- Nutritional or feeding systems
- Infusion-related assemblies
- Veterinary surgical setups
- Laboratory circulation systems
Each of these environments involves periodic manipulation of connectors. Antimicrobial materials reduce microbial persistence on external and internal surfaces following handling events.
Closed-system design principles still apply. Connector antimicrobial properties support, but do not replace, proper aseptic technique.
Material science considerations behind anti microbial polymers
Anti microbial performance is engineered through chemical and structural modification of base polymers. Silver-based additives, quaternary ammonium compounds, and organic antimicrobial agents are commonly used in polymer integration.
Two integration methods dominate:
- Compounded integration where antimicrobial agents are mixed into polymer pellets before extrusion or molding
- Surface bonding or coating applied after component fabrication
Compounded integration offers uniform distribution throughout the material cross-section. Surface treatments provide targeted exposure but may degrade under abrasion.
Material selection must also address:
- Extractables and leachables
- Fluid compatibility
- Optical clarity requirements
- Sterilization method compatibility
In medical and veterinary applications, validation extends beyond antimicrobial performance to include biocompatibility and mechanical reliability.
Design tradeoffs when specifying anti microbial tubing systems
Anti microbial tubing introduces tradeoffs that must be evaluated at the system level. Cost, manufacturability, and regulatory documentation requirements increase with material complexity.
Decision factors include:
- Duration of use
- Risk tolerance
- Sterilization protocol
- Mechanical stress profile
- Regulatory classification
Single-use disposable systems may benefit from integrated antimicrobial materials without requiring long-term durability. Reusable systems must evaluate surface degradation over multiple cleaning cycles.
Connector integration must also consider torque resistance, thread integrity, and sealing reliability. Antimicrobial additives should not compromise mechanical strength.
Performance durability over time
Anti microbial performance diminishes if active agents migrate out of the polymer or if surface treatments erode. Longevity depends on additive chemistry and environmental stressors.
Durability factors include:
- Repeated sterilization cycles
- UV exposure
- Mechanical abrasion
- Fluid chemistry
- Temperature extremes
In high-temperature sterilization environments, certain antimicrobial compounds degrade. Ethylene oxide or low-temperature methods may preserve surface integrity more effectively.
Evaluation should include lifecycle testing rather than initial antimicrobial validation alone.
Integration into veterinary and laboratory environments
Veterinary and laboratory settings present unique contamination patterns that differ from acute human healthcare environments. Organic matter variability, animal handling conditions, and multi-use spaces introduce unpredictable microbial loads.
Anti microbial tubing and connectors support:
- Sample integrity
- Reduced cross-transfer risk
- Cleaner fluid transport pathways
- Enhanced environmental resilience
Component sourcing from specialized suppliers with domain-specific expertise ensures compatibility with veterinary equipment and laboratory instrumentation. BOMImed supports clinical and veterinary facilities with equipment and components aligned to these operational realities, including fluid management systems that prioritize durability and contamination mitigation.
Material transparency and chemical compatibility remain essential in these environments, particularly when monitoring fluid characteristics or administering treatments.
Regulatory and compliance alignment
Anti microbial claims must align with applicable regulatory standards governing medical devices and laboratory equipment. Overstated antimicrobial claims can create compliance exposure.
Manufacturers and distributors should document:
- Antimicrobial testing methodology
- Duration of efficacy
- Material composition
- Biocompatibility validation
Claims should reflect measurable performance under defined test conditions. Regulatory review may differ between jurisdictions, and labeling language must remain precise.
Distributors serving healthcare and veterinary markets must ensure documentation clarity when specifying anti microbial tubing and connectors.
Cost implications and procurement strategy
Anti microbial components typically carry a higher unit cost due to material additives and validation processes. Procurement decisions should evaluate total system risk rather than unit price alone.
Cost considerations include:
- Reduced contamination incidents
- Lower cleaning burden
- Decreased downtime
- Improved system reliability
High-risk applications often justify the incremental cost through risk mitigation value. In low-exposure systems, standard tubing may remain appropriate.
Procurement strategy should be driven by exposure level, handling frequency, and environmental conditions rather than blanket standardization.
Anti Microbial Tubing & Connectors – People Also Ask
What is anti microbial tubing used for?
Anti microbial tubing is used in fluid transport systems where contamination risk must be minimized, including medical, veterinary, and laboratory applications.
Does anti microbial tubing replace sterilization?
Anti microbial tubing does not replace sterilization. It supplements hygiene protocols by reducing microbial growth between cleaning or sterilization cycles.
How do anti microbial connectors work?
Anti microbial connectors incorporate materials or surface treatments that inhibit microbial survival at connection points, reducing contamination risk during handling.
Are anti microbial materials safe for medical use?
Anti microbial materials are safe when properly validated for biocompatibility and regulatory compliance in their intended application.
Do anti microbial additives wear out over time?
Anti microbial performance can diminish due to surface wear, sterilization exposure, or chemical degradation, depending on the material formulation.
Is anti microbial tubing necessary for all fluid systems?
Anti microbial tubing is most beneficial in systems with repeated handling or high contamination exposure; not all fluid systems require it.
Can anti microbial tubing prevent biofilm completely?
Anti microbial tubing reduces biofilm formation risk but does not guarantee complete prevention under all environmental conditions.